How Can Habitat Fragmentation Contribute to Biodiversity Loss?
Habitat loss and fragmentation has long been considered the principal cause for biodiversity loss and ecosystem degradation worldwide, and is a key inquiry topic in landscape ecology (Wu 2013). Habitat fragmentation often refers to the reduction of continuous tracts of habitat to smaller, spatially distinct remnant patches, and habitat loss typically occurs concurrently with habitat fragmentation (Collinge 2009). Although some habitats are naturally patchy in terms of abiotic and biotic conditions (Wu and Loucks 1995), homo actions have profoundly fragmented landscapes across the word (Haddad et al. 2015), altering the quality and connectivity of habitats. Therefore, understanding the causes and consequences of habitat fragmentation is critical to preserving biodiversity and ecosystem functioning.
From May fourth to 10th, 2015, an International Workshop on Habitat Fragmentation and Biodiversity Conservation, held at the One thousand Island Lake, Zhejiang, China, discussed threats to biodiversity in fragmented landscapes and how fragmentation research can identify and help mitigate these threats. To meet these challenges, the Workshop had 3 goals. The first was to synthesize key findings in fragmentation science. Second was to place important remaining inquiry questions concerning the relationships between habitat fragmentation, biodiversity, and ecosystem functioning at local, regional, and global scales. Finally, we examined the unique roles of field-based fragmentation experiments in addressing these questions. The Workshop'south findings are relevant to the broader ecological community, and we present them hither to stimulate research that will advance landscape ecology and conservation biology.
Key findings concerning habitat loss and fragmentation
-
While habitat fragmentation ultimately derives from habitat loss, three broadly defined mechanisms mediate the ecological consequences of fragmentation. First, there are those attributable directly to the loss of habitat area. Second, there are those attributable directly to changes in the spatial configuration of the landscape, such every bit isolation. Finally, there are those owing to indirect or interaction effects of habitat loss and changes in spatial configuration (Didham et al. 2012), and to the interaction of fragments with the matrix (east.chiliad., spillover furnishings). A review of the literature found that when i ignores indirect and interaction effects, the impacts of habitat loss are far greater than changing habitat configuration (Fahrig 2003); however, newer research suggests that indirect and interaction effects may be the dominant commuter of the ecological changes oftentimes attributed to habitat loss solitary (Didham et al. 2012).
-
Species richness often changes significantly with fragmentation (MacArthur and Wilson 1967; Diamond 1975). Nonetheless, other measures of community structure, such as community limerick, trophic organization, species persistence, and species residency, may better inform how fragmentation affects biotic communities, even when species richness per se is non altered past fragmentation (Robinson et al. 1992; Haddad et al. 2015).
-
Every bit habitat loss results in changes in both the amount and configuration of habitat (east.g., decreased patch size, increased patch isolation, and increased edge area), fragmentation-mediated processes cause generalizable responses at the population, community, and ecosystem levels. These include decreased residency within and movement amidst fragments, reduced species richness across taxonomic groups, and decreased nutrient retention (Haddad et al. 2015). By comparing findings beyond multiple landscape-scale fragmentation experiments, one can partition effectively the relative influence of increasing habitat loss, patch isolation, and edge influence on different community and ecosystem attributes (Haddad et al. 2015), and potentially distinguish generalizable consequences of fragmentation from more idiosyncratic, system-specific responses.
-
Area and isolation furnishings encompass a diverseness of ecological processes that can complicate our understanding of fragmentation. For example, reductions in patch size and increases in edge-affected area can influence local ecosystem processes indirectly through microclimatic furnishings. To make results more generalizable, studies should decompose expanse and isolation effects into direct, ecologically relevant, mechanistic drivers such as microclimate, local matrix quality, and vulnerability to stochastic events (e.1000., Laurance et al. 2011).
Remaining questions and challenges
Despite the progress made in formalizing fragmentation science, significant questions remain.
Local community-level dynamics
How do atmospheric condition at the time of fragmentation impact community structure and dynamics?
Historically the term fragmentation has been used to describe the ecological changes arising from two different landscape contexts. The first of these are relaxing systems—those intact at the time of fragmentation and which are now relaxing to few species and macerated ecosystem function (e.1000., Laurance et al. 2011). In contrast, assembling systems are those in which successional processes occur inside spatially singled-out patches across a denuded mural (due east.yard., Cook et al. 2005). Most studies have focused on communities that were intact at the time of fragmentation, losing species following the fragmentation event. Relatively few studies have focused on the impacts of habitat fragmentation on community associates (east.g., Simberloff and Wilson 1970; Robinson et al. 1992). For instance, priority effects (Fukami 2015) and a combination of deterministic and stochastic processes may influence the trajectory of community assembly in fragmented systems (Norden et al. 2015). Patterns of beta diversity can be contradistinct by fragmentation overlaying successional dynamics (Alexander et al. 2012). While the processes of assembly and relaxation practice share some generalizable effects (Haddad et al. 2015, discussed above), nosotros exercise not fully empathise the ways in which these systems converge and diverge from each other, creating a primary source of confusion inside the study of fragmentation.
What factors touch the relative rest between top-downward and bottom-up processes in fragmented landscapes?
Customs ecologists have long recognized that both top–down (predator–mediated) and bottom–up (producer–mediated) processes can influence community limerick. Notwithstanding, our understanding of how top-downwardly and bottom-upwardly dynamics might interact across geographic infinite is limited (Gripenberg and Roslin 2007), although information technology is clear that spatial factors such as patch size could potentially impact the strength of trophic cascades (Terborgh 2010). Fragmented habitats present an excellent platform for examining the interaction between these forces over a variety of spatial scales.
How practice the processes of dispersal and ecological filtering—exclusion due to the effects of ecology and biotic conditions—interact to structure biodiversity?
Although both dispersal- and niche-mediated mechanisms affect community assembly, the importance of the interaction between these processes in high-diversity communities is still largely unknown (Myers and Harms 2011). Fragmentation studies can be used to address this outcome every bit well, especially when fragmentation eliminates some of the key players in biotic interactions such as mutualism and competition.
How does fragmentation predictably and consistently alter the variability of local biophysical conditions?
Big-scale experiments show that fragmentation has significant, unexpected furnishings on local biophysical conditions. For example, the Biological Dynamics of Woods Fragments Project of Manaus, Brazil finds that relatively unimportant drivers in intact systems can come to dominate fragmented systems. Examples include increased wind shear in tropical wood fragments (Laurance et al. 2011), and reduced fire frequency, in prairie systems (Leach and Givnish 1996). We need further work on contradistinct disturbance regimes, peculiarly focusing on the integration of patch shape and matrix characteristics, to generalize these results across landscapes and disturbance regimes. The inclusion on micro-climatic changes in studies of fragmented landscapes may too provide new insights into the mechanisms behind changes in community composition.
How rapidly does evolutionary adaptation in fragmented habitats occur? How does this evolutionary adaption touch on species coexistence and community associates in fragmented habitats?
Recent work has shown that evolutionary changes can operate on ecological fourth dimension scales. Local adaptations, whether in the form of evolutionary change or phenotypic plasticity, may change the environmental selective pressures that control community assembly (Schoener 2011). Fragmentation research should play a key role in quantifying the speed of this adaptation to local conditions and how these adaptations alter species coexistence and community assembly.
Do smaller patches become "stuck" in successional stasis? If then, does this successional heterogeneity between larger and smaller patches alter the power of biodiversity to recover from farther perturbations at the mural scale?
From the early twentieth century, ecologists accept suspected that habitat fragmentation can alter successional trajectories, with smaller patches affected more than profoundly than larger ones (Clements 1936). Cook et al. (2005) report that succession towards woody vegetation occurs more slowly on pocket-size fragments, and Connell and Slatyer (1977) advise that succession should reflect the interaction of disturbance intensity and size of the area beingness disturbed. Yet understanding the relationship betwixt fragmentation and succession is largely unexplored.
How quickly and effectively can management actions mitigate declining numbers of species and diminished ecosystem functions post-obit fragmentation?
The effects of habitat fragmentation on populations, communities, and ecosystems can take years to decades earlier becoming credible, suggesting that patches will continue to lose species and see declines in ecosystem functions for considerable fourth dimension periods (e.g., Brooks et al. 1999; Collins et al. 2009). Our ability to reduce the loss of species and functions during this flow of decline merits investigation.
How does fragmentation touch on population dynamics at the species level? How do these dynamics alter the relative probabilities of extirpations and recolonization?
Though traditional island biogeography is based on the probability of species extirpations and colonizations, it does not make predictions nigh how habitat fragmentation will affect the population dynamics of individual species. Therefore, metapopulation models based upon occupancy (presence/absences) take provided the frameworks generally used to study the impacts of fragmentation at the species level. From their very inception (Levins 1969, 1970), these frameworks have largely ignored the details of intra-patch population dynamics (Gilpin and Hanski 1991). Yet, such local population dynamics underlie the local extirpations and (to a bottom extent) recolonizations of individual species within and amidst patches. Thus, they correspond a critical, if largely unstudied, component in applying fragmentation research at the species level. For instance, do time-series of abundances for a focal species typical prove clear signals of population decline in fragments, long before extinction actually occurs?
Landscape-scale dynamics
How do border contrast and matrix quality change metacommunity dynamics?
The relative similarity or contrast between habitat and non-habitat areas ("border contrast") and matrix quality in an absolute sense can affect both individual organisms and material fluxes (Ricketts 2001; Prevedelo and Vieira 2010). Further, fragmented landscapes tin can allow for the persistence of a metacommunity, or a set up of local communities that are connected through the processes of dispersal and extirpation (Gilpin and Hanski 1991; Wilson 1992; Holyoak et al. 2005). Studies have not considered how the interaction of these mechanisms affects metacommunity dynamics in terms of patch-specific dispersal, colonization, and extirpation probabilities of species, especially in the context of fragmentation.
How does habitat fragmentation alter relationships between biodiversity and ecosystem functioning across multiple scales?
There is wide recognition that fragmentation affects both biodiversity and ecosystem functioning (Haddad et al. 2015). Our noesis of biodiversity-ecosystem performance relationships suggests that local loss of biodiversity causes a local loss of function. These expectations extrapolate from a enquiry base in which community biodiversity was experimentally altered in a randomized blueprint (Loreau et al. 2001; Hooper et al. 2005), rather than altered by the non-random species losses that fragmentation causes (e.m., Ewers and Didham 2006). The furnishings of fragmentation on relationships betwixt biodiversity and functioning merit further study.
How will the local microclimatic furnishings of fragmentation interact with climate change on the individual, population, customs, ecosystem, and landscape levels?
Fragmentation can change the microclimate at both local and regional scales (eastward.m., Young and Mithchell 1994; Didham and Lawton 1999; Laurance et al. 2011). These fragmentation-based climatic changes may interact with other types of anthropogenic climate alter, farther influencing biodiversity through unstudied interactions betwixt the ii drivers.
What is the appropriate spatial scale for studying the ecological impacts more often than not attributed to habitat fragmentation?
The ecological impacts of fragmentation occur across a multifariousness of spatial scales. For example, edge effects affect populations and communities on within-patch scales. Dispersal acts on both within-patch and between-patch scales, and changes in ecosystem services occur beyond multiple scales. The literature does not well constitute these scaling relationships equally fragmentation studies have rarely taken a hierarchical, multiple-scale approach (Didham et al. 2012).
The role of fragmentation experiments
Field-based fragmentation experiments are disquisitional in expanding our understanding of habitat fragmentation. Ranging in spatial scale from two × 10−vii ha to 100 ha, the virtually commonly recognized fragmentation experiments cover a broad range of ecological communities (Fig. one; Haddad et al. 2015). In contrast to observational studies, these projects have careful, a priori, experimental designs with pregnant levels of replication and known initial weather, allowing for powerful inferences. Few even so approach the scale at which gimmicky land management and conservation planning must accost fragmentation.
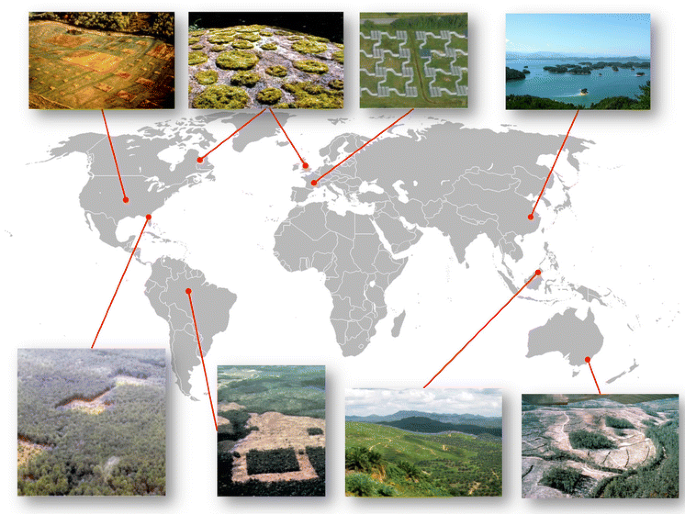
Map of long-term fragmentation experiments as identified in Haddad et al. (2015) with the improver of Thousand Isle Lake, clockwise from summit-left: 1 Kansas Fragmentation Project (KFP). Located in Kansas, USA, KFP is an experimentally fragmented prairie ecosystem, focusing on the impacts of fragmentation on community associates and successional processes. two Moss fragmentation experiments (MFE). Consisting of a wide range of projects carried out simultaneously in both the Great britain and Canada, MFE includes both field and laboratory experiments, which have focused on a broad assortment of processes, including fragmentation per se, corridor effectiveness, and the interactions between fragmentation and climate alter. 3 Metatron. Possibly the most technically complex and flexible fragmentation experiment, Metatron, located in south central France, consists of independent patches which tin can be connected or disconnected via experimentally controlled corridors, allowing for the written report of multiple landscape configurations. iv Thousand Island Lake (TIL). Described in particular in the text, TIL combines both customs associates and relaxation processes across more than thousand remnant islands. five Wog Wog Habitat Fragmentation Experiment (WWHFE). Located in Southeastern Commonwealth of australia, WWHFE was designed to study the effects of habitat fragmentation on biological variety in an Eucalyptus wood. 6 Stability of Altered Forest Ecosystems (SAFE) Project. Located in the rainforests of Malaysian Borneo, SAFE is composed of multiple projects that investigate how forest modification gradients (e.g., land apply and comprehend patterns) and forest fragmentation affect biodiversity, ecological processes, and waterways. 7 Biological Dynamics of Wood Fragments Project (BDFFP). Located in the Brazilian Amazon, BDFFP is the world's largest and longest-running habitat fragmentation experiment, conducting a wide range of wood fragmentation effects on biodiversity and ecosystem processes. eight Savannah River Site Corridor Experiment (SRSCE). Located in South Carolina, United states, SRSCE was designed to study the effects of corridors on plant and animal dispersal, population persistence, and biodiversity in a managed woods
One challenge is to bridge the gap between scales feasible for straight experimental manipulation, and larger scales which are the domain of conservation, restoration, and management. Large-scale infrastructure projects can at times provide inadvertent, unplanned experiments which can exist utilized to fill this gap in scales. For example, in Venezuela, Professor John Terborgh creatively utilized the creation of Lago Guri, a large (>4000 kmtwo) man-made hydroelectric lake dotted with hundreds of forested islands, to demonstrate the dramatic importance of trophic cascades in tropical wood (Terborgh et al. 2001). Unfortunately, the draining of the lake destroyed the integrity of the study, and combined with political uncertainties, research there is not ongoing.
Another promising large-calibration, unplanned experiment, comparable in some ways to Lago Guri, which tin fill the gap between standard fragmentation experiments and large spatial scales is the ongoing projection at One thousand Isle Lake (TIL). Formed in 1959 TIL is a large, homo-made lake in Chun'an County of Zhejiang Province, China. TIL has total h2o surface of approximately 580 km2 and 1078 land-bridge islands when the maximum h2o level (108 k.a.southward.50.) (Figure 1; Wang et al. 2009). During dam construction, principal forests in the region were selectively or clear-cut with organized logging during the "Not bad Jump Frontwards." This resulted in nearly complete deforestation before the lake's inundation. Airplanes then sowed native pines, maybe affecting soil pH or causing allelopathy that could accept lasting effects on regional biotas. This region is at present protected as i of the largest national parks in China. The bulk of the islands (sometime hilltops) take not experienced significant homo disturbance since 1962. In concert, these reasonably consistent initial conditions combined with a history of ecological monitoring and the opportunity for very loftier levels of replication, allow the TIL system to avert many of the pitfalls that plague other "natural" fragmentation experiments. This history besides ensures that TIL volition combine aspects of relaxing and assembling ecosystems, in that the original understory community was not directly removed, and the removal of copse would set into move successional dynamics equally recolonization occurs from external seed sources.
Studies at TIL have already provided the building blocks necessary to answer many of the questions proposed in a higher place, providing insights into the recovery patterns of avian, mammalian, reptilian, and constitute communities in fragmented landscapes. A suite of studies, focusing on both institute and animal communities, have shown that island expanse plays the ascendant role in controlling community recovery on TIL's islands. On the species and factor levels, plant, bird, snake, and small mammal communities have clear relationships with island surface area, simply not island isolation (Wang et al. 2010, 2011, 2012b; Hu et al. 2011; Zhang et al. 2012; Ding et al. 2013; Si et al. 2014, 2015a; Su et al. 2014; Yuan et al. 2015). There are some notable exceptions to the trend of expanse effects dominating isolation effects (e.g., Wang et al. 2012a; Yu et al. 2012; Peng et al. 2014). That said, when taken as a whole these results suggest that patch expanse may be a more than significant cistron in community assembly than patch isolation in this system. Ongoing research is underway to dissect the specific mechanisms past which this process occurs (e.g., Hu et al. 2015; Si et al. 2015b).
The opportunity for comparing between TIL to other large scale fragmentation experiments is promising. Because the communities of TIL are primarily assembling, straight comparisons to other assembling communities such as the Kansas Fragmentation Experiment (east.g., Cook et al. 2005) could provide insights into whether successional processes in fragmented landscapes are consistent beyond biomes. Used together, systems in which matrix quality remains abiding can compare to large-scale experiments in which matrix quality varies, such as the Biological Dynamics of Forest Fragments Projection (east.thou., Laurance et al. 2011), to address questions concerning edge contrast. Naturally, there are many other opportunities for comparison. We wait the TIL project to play a critical function in fragmentation research over the coming decades, while also proving a platform for international collaborations.
Fragmentation studies in the past take enabled u.s.a. to sympathise a broad range of furnishings of habitat loss and fragmentation on biodiversity and ecological processes, but primal questions remain. These questions span spatial, temporal, and organizational scales, and they necessitate new approaches and techniques. With a focus on identifying ecologically relevant drivers, we are confident that answering these questions will provide scientists and practitioners with the scientific basis and tools necessary to promote biodiversity and mural sustainability.
References
-
Alexander HM, Foster BL, Ballantyne F IV, Collins CD, Antonovics J, Holt RD (2012) Metapopulations and metacommunities: combining spatial and temporal perspectives in institute ecology. J Ecol 100:88–103
-
Brooks TM, Pimm SL, Oyugi JO (1999) Time lag between deforestation and bird extinction in Tropical Woods Fragments. Conserv Biol 13:1140–1150
-
Clements FE (1936) Nature and construction of the climax. J Ecol 24:252–284
-
Collinge SK (2009) Ecology of fragmented landscapes. Johns Hopkins University Press, Baltimore
-
Collins CD, Holt RD, Foster BL (2009) Patch size effects on plant species decline in an experimentally fragmented landscape. Ecology 90:2577–2588
-
Connell JH, Slatyer RO (1977) Mechanisms of succession in natural communities and their role in community stability and organization. Am Nat 111:1119–1144
-
Cook WM, Yao J, Foster BL, Holt RD, Patrick LB (2005) Secondary succession in an experimentally fragmented landscape: community patterns across space and time. Environmental 86:1267–1279
-
Diamond JM (1975) The isle dilemma: lessons of modern biogeographic studies for the pattern of natural reserves. Biol Conserv 7:129–146
-
Didham RK, Lawton JH (1999) Edge structure determines the magnitude of changes in microclimate and vegetation construction in tropical wood fragments. Biotropica 31:17–xxx
-
Didham RK, Kapos V, Ewers RM (2012) Rethinking the conceptual foundations of habitat fragmentation inquiry. Oikos 121:161–170
-
Ding Z, Feeley KJ, Wang Y, Pakeman RJ, Ding P (2013) Patterns of bird functional diversity on land-span isle fragments. J Anim Ecol 82:781–790
-
Ewers RM, Didham RK (2006) Misreckoning factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117–142
-
Fahrig Fifty (2003) Effects of habitat fragmentation on biodiversity. Ann Rev Ecol Syst 34:487–515
-
Fukami T (2015) Historical contingency in community assembly: integrating niches, species pools, and priority effects. Ann Rev Ecol Evol Syst 46:i–23
-
Gilpin ME, Hanski IA (1991) Metapopulation dynamics: empirical and theoretical investigations. Academic Printing, London
-
Gripenberg S, Roslin T (2007) Upwardly or downwards in space? Uniting the lesser-upward versus acme-downward epitome and spatial environmental. Oikos 116:181–188
-
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Collins CD, Cook WM, Damschen EI, Ewers RM, Foster BL, Jenkins CN, Male monarch AJ, Laurance WF, Levey DJ, Margules CR, Melbourne BA, Nicholls AO, Orrock JL, Song DX, Townshend JR (2015) Habitat fragmentation and its lasting touch on Earth's ecosystems. Sci Adv 1:e1500052
-
Holyoak M, Leibold MA, Holt RD (2005) Metacommunities: spatial dynamics and ecological communities. University of Chicago Press, Chicago, pp 1–31
-
Hooper DU, Chapin FS III, Ewel JJ, Hector A, Inchausti P, Lavorel South, Lawton JH, Club DM, Loreau M, Naeem S, Schmid B, Setälä H, Symstad AJ, Vandermeer J, Wardle DA (2005) Effects of biodiversity on ecosystem functioning: a consensus of current noesis. Ecol Monogr 75:3–35
-
Hu G, Feely KJ, Wu J, Xu GF, Yu MJ (2011) Determinants of plant species richness and patterns of nestedness in fragmented landscapes: testify from state-bridge islands. Landsc Ecol 26:1405–1417
-
Hu 1000, Feeley KJ, Yu 1000 (2015) Configuration of fragmented habitat drives plant community assembly processes across plant life stages. J Biogeogr (in review)
-
Laurance WF, Camargo JLC, Luizao RCC, Laurance SG, Pimm SL, Bruna EM, Stouffer PC, Williamson GB, Benitez-Malvido J, Vasconcelos HL, Van Houtan KS, Zartman CE, Boyle SA, Didham RK, Andrade A, Lovejoy TE (2011) The fate of Amazonian forest fragments: a 32-year investigation. Biol Conserv 144:56–67
-
Leach MK, Givnish TJ (1996) Ecological determinates of species loss in remnant prairies. Science 237:1555–1558
-
Levins R (1969) Some demographic and genetic consequences of ecology heterogeneity for biological control. Bul Entomol Soc Am 15:237–240
-
Levins R (1970) Extinction. In: Gesternhaber Chiliad (ed) Some mathematical problems in biological science. American Mathematical Society, Providence, pp 77–107
-
Loreau Thou, Naeem S, Inchausti P, Bengtsson J, Grime JP, Hector A, Hooper DU, Huston MA, Raffaelli D, Schmid B, Tilman D, Wardle DA (2001) Biodiversity and ecosystem functioning: current noesis and future challenges. Scientific discipline 294:804–808
-
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton Academy Press, Princeton
-
Myers JA, Harms KE (2011) Seed inflow and ecological filters interact to assemble loftier-diverseness plant communities. Ecology 92:676–686
-
Norden N, Angarita HA, Bongers F, Martínez-Ramos 1000, Granzow-de la Cerda I, van Breugel 1000, Lebrija-Trejos E, Meave JA, Vandermeer J, Williamson GB, Finegan B, Mesquita R, Chazdon RL (2015) Successional dynamics in neotropical forests are as uncertain as they are predictable. Proc Natl Acad Sci USA 112:8013–8018
-
Peng S, Hu G, Yu Chiliad (2014) Beta diversity of vascular plants and its influencing factors on islands in the One thousand Isle Lake. Acta Ecol Sinica 34:3866–3872 (in Chinese with English abstract)
-
Prevedelo JA, Vieira MV (2010) Does the blazon of matrix matter? A quantitative review of the evidence. Biodivers Conserv 19:1205–1223
-
Ricketts TH (2001) The matrix matters: constructive isolation in fragmented landscapes. Am Nat 158:87–99
-
Robinson GR, Holt RD, Gaines MS, Hamburg SP, Johnson ML, Fitch HS, Martinko EA (1992) Diverse and contrasting furnishings of habitat fragmentation. Scientific discipline 257:524–526
-
Schoener TW (2011) The newest synthesis: agreement the interplay of evolutionary and ecological dynamics. Science 331:426–429
-
Si X, Pimm SL, Russell GJ, Ding P (2014) Turnover of breeding bird communities on islands in an inundated lake. J Biogeogr 41:2283–2292
-
Si X, Baselga A, Ding P (2015a) Revealing beta-variety patterns of breeding bird and lizard communities on inundated land-span islands past separating the turnover and nestedness components. PLoS I 10:e0127692
-
Si 10, Baselga A, Leprieur F, Song X, Ding P (2015b) Selective extinction drives taxonomic and functional alpha and beta diversities in island bird assemblages. J Anim Ecol (accepted)
-
Simberloff DS, Wilson EO (1970) Experimental zoogeography of islands: a two year record of colonization. Environmental 51:934–937
-
Su X, Yuan J, Hu 1000, Xu G, Yu M (2014) Edge effect of the institute community structure on country-bridge islands in the Thousand Island Lake. Mentum J Appl Ecol 25:77–84 (in Chinese with English abstract)
-
Terborgh J (2010) The trophic cascade on islands. In: Losos JB, Ricklefs RE (eds) The theory of island biogeography revisited. Princeton University Printing, Princeton, pp 116–142
-
Terborgh J, Lopez Fifty, Nuñez P, Rao Yard, Shahabuddin G, Orihuela G, Riveros M, Ascanio R, Adler GH, Lambert TD, Balbas L (2001) Ecological meltdown in predator-complimentary forest fragments. Science 294:1923
-
Wang Y, Zhang J, Feeley KJ, Jiang P, Ding P (2009) Life-history traits associated with fragmentation vulnerability of lizards in the Thousand Island Lake, China. Anim Conserv 12:329–337
-
Wang Y, Bao Y, Yu Yard, Xu G, Ding P (2010) Nestedness for unlike reasons: the distributions of birds, lizards and small mammals on islands of an inundated lake. Divers Distrib 16:862–873
-
Wang R, Compton SG, Chen XY (2011) Fragmentation can increase spatial genetic construction without decreasing pollen-mediated gene flow in a wind-pollinated tree. Mol Ecol 20:4421–4432
-
Wang Y, Lane A, Ding P (2012a) Sex-biased dispersal of a frog (Odorrana schmackeri) is affected by patch isolation and resource limitation in a fragmented landscape. PLoS Ane 7(10):e47683
-
Wang Y, Wang 10, Ding P (2012b) Nestedness of snake assemblages on islands of an inundated lake. Curr Zool 58:828–836
-
Wilson DS (1992) Complex interactions in metacommunities, with implications for biodiversity and college levels of pick. Ecololgy 73:1984–2000
-
Wu JG (2013) Primal concepts and research topics in landscape environmental revisited: xxx years after the Allerton Park workshop. Landsc Ecol 28:i–eleven
-
Wu JG, Loucks OL (1995) From balance of nature to hierarchical patch dynamics: a paradigm shift in environmental. Q Rev Biol lxx:439–466
-
Immature A, Mithchell N (1994) Microclimate and vegetation edge furnishings in a fragmented podocarp-broadleaf forest in New Zealand. Biol Conserv 67:63–72
-
Yu M, Hu G, Feeley KJ, Wu J, Ding P (2012) Richness and limerick of plants and birds on country-span islands: effects of isle attributes and differential responses of species groups. J Biogeogr 39:1124–1133
-
Yuan N, Comes HP, Cao YN, Guo R, Zhang YH, Qiu YX (2015) A comparative study on genetic furnishings of artificial and natural habitat fragmentation on Loropetalum chinense (Hamamelidaceae) in Southeast China. Heredity 114:544–551
-
Zhang X, Shi MM, Shen DW, Chen XY (2012) Habitat loss other than fragmentation per se decreased nuclear and chloroplast genetic diversity in a monoecious tree. PLoS Ane seven:e39146
Acknowledgments
The workshop was funded past the Zhejiang Clan for Scientific discipline and Technology, the Ecological Society of Zhejiang Province, the Botanical Order of Zhejiang Province, the People'southward Government of Chun'an County, the Yard Island Lake National Forest Park of Zhejiang and the National Natural Science Foundation of Prc (Grant Number 31210103908). National Natural Scientific discipline Foundation of China (Grant Numbers 31210103908, 31361123001 and 31200413) and the US National Scientific discipline Foundation (Grant Numbers DEB-1342754 and DEB-1342757) have too supported the projects in the Thousand Island Lake.
Author data
Affiliations
Corresponding authors
Rights and permissions
Well-nigh this article
Cite this article
Wilson, M.C., Chen, XY., Corlett, R.T. et al. Habitat fragmentation and biodiversity conservation: key findings and future challenges. Landscape Ecol 31, 219–227 (2016). https://doi.org/10.1007/s10980-015-0312-3
-
Received:
-
Accustomed:
-
Published:
-
Issue Appointment:
-
DOI : https://doi.org/ten.1007/s10980-015-0312-3
Source: https://link.springer.com/article/10.1007/s10980-015-0312-3
0 Response to "How Can Habitat Fragmentation Contribute to Biodiversity Loss?"
Post a Comment